ORIGIN
Patient Information
What is brachytherapy?
Radiotherapy is the use of ionizing radiation for the treatment of cancer and 50 – 60 % of patients require radiotherapy at some point during their treatment[1]. It is delivered in the form of external beam radiotherapy (EBRT) using linear accelerators (linacs), or internally, known as brachytherapy (BT).
Brachytherapy is further divided into Low Dose Rate (LDR), where the radioactive sources, known as seeds, remain implanted permanently, and High Dose Rate (HDR), where higher activity radiation seeds, with dose rates > 12Gy/hr, are temporarily implanted.
- In the case of LDR-BT (see figure 1a), these seeds are usually Iodine-125, with apparent activities of the individual seeds typically between 0.357mCi and 0.42mCi respectively, and have a half-life of 59.43 days.
- In HDR-BT (see figure 1b), a computer driven device, known as a remote afterloader, pushes the radioactive source, typically Iridium-192 or Cobalt-60, which is attached to a wire, through the applicators to the treatment site. The radioactive source is moved in millimetre increments and remains in a certain position (dwell position) for a predetermined amount of time. Once the planned treatment dose has been achieved, the remote afterloader automatically withdraws the radioactive source so there is no residual radiation or radioactivity in the patient. Each treatment typically takes 10 – 15 minutes and is delivered in a number of sessions, known as fractions. In cervical cancer for example, a patient would typically receive 1 – 10 fractions of 3 – 20 Gy depending on the indication.
[1] Rosenblatt, E., Acuña, O., & Abdel-Wahab, M. (2015). The Challenge of Global Radiation Therapy: An IAEA Perspective. International Journal of Radiation Oncology• Biology• Physics, 91(4), 687-689.
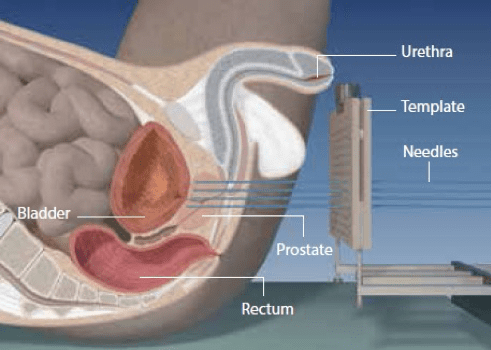
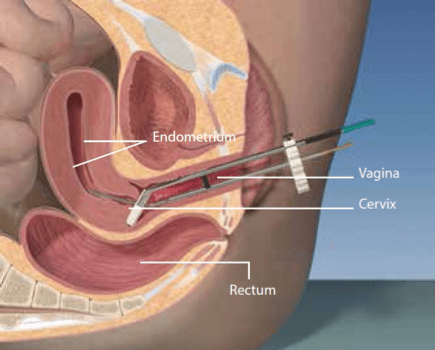
Figure 1. Current brachytherapy delivery devices for prostate (left) and cervical (right) cancers [Eckert & Ziegler BEBIG]
LDR Brachytherapy treatment of prostate cancer [Eckert & Ziegler BEBIG]
Correct placement of the radiation source is vital to ensure adequate radiation to the target area (tumour), while ensuring minimum exposure to nearby critical organs, such as, in the case of prostate and gynaecological cancers, the bladder, urethra and rectal wall.
Monitoring the precise radiation dose delivered to critical organs near the tumour, such as the urethra and rectal wall, is crucial to reduce these adverse side-effects and further improve the quality of life of cancer survivors.
How will ORIGIN improve brachytherapy?
ORIGIN aims to deliver more effective, photonics-enabled, brachytherapy for cancer treatment through advanced real-time radiation dose imaging and source localisation. This will be achieved by the development of a new optical fibre based sensor system to support diagnostics-driven therapy through enhanced adaptive brachytherapy.
ORIGIN brings together a highly multidisciplinary consortium of leaders in their respective fields (academia and industry with photonics, engineering, medical physics, radiobiology, and clinical expertise), in order to develop a new 16 point array optical fibre dosimeter for both LDR- and HDR-BT, with novel algorithms to provide 3D dose imaging with source localisation capability. The involvement of both industry and clinical partners from the outset will ensure that patient care is at the forefront across the entire value chain. The integration of the ORIGIN system within existing clinical treatment planning systems and after-loader delivery devices will provide enhanced adaptive therapy, for improved patient outcomes.
Read about the latest research, news and activities within the ORIGIN project in our newsletters!








